Address
304 North Cardinal St.
Dorchester Center, MA 02124
Work Hours
Monday to Friday: 7AM - 7PM
Weekend: 10AM - 5PM
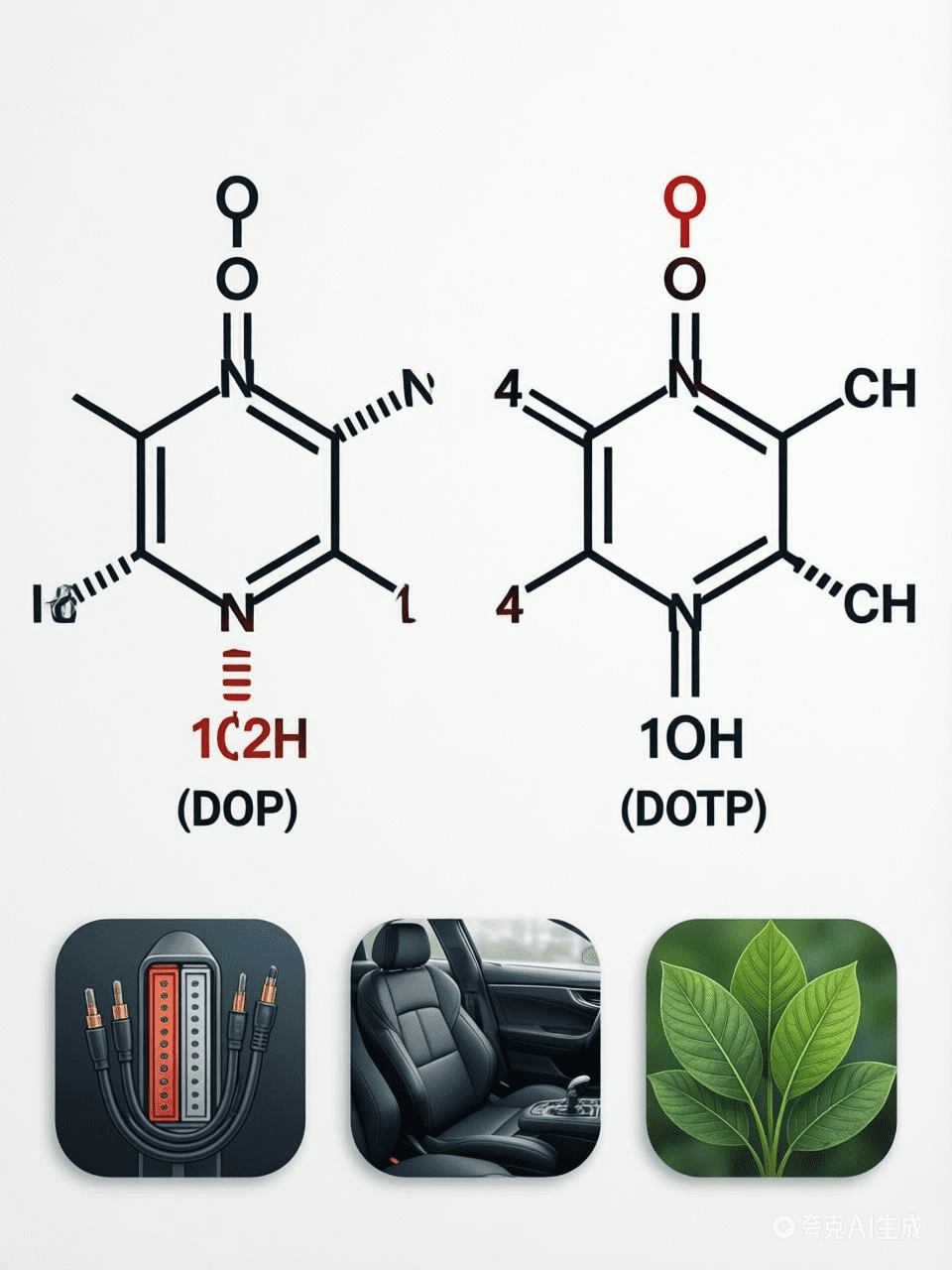
Among the most common are Di-Octyl Phthalate (DOP) and Di-Octyl Terephthalate (DOTP).
In the vast world of polymer science, plasticizers play a crucial, albeit often invisible, role in shaping the materials we use daily. Among the most common are Di-Octyl Phthalate (DOP) and Di-Octyl Terephthalate (DOTP). While both serve the primary function of increasing the plasticity of materials like Polyvinyl Chloride (PVC), their chemical structures, performance, and environmental footprints differ significantly. This article explores the distinctions between these two compounds, highlighting why DOTP is increasingly becoming the preferred choice in a sustainability-conscious market.


Chemical Structure and Origin
At the molecular level, the difference begins with the acid used in their synthesis. DOP is derived from ortho-phthalic acid, while DOTP is synthesized from terephthalic acid. This subtle shift in the position of the carboxylic acid groups on the benzene ring—from adjacent positions in DOP to opposite positions in DOTP—grants DOTP a more linear molecular structure. This linearity is a key factor in DOTP’s superior compatibility with PVC matrices and its enhanced performance characteristics.
Performance Comparison
When comparing the two plasticizers, several critical parameters stand out:
Environmental and Safety Profile
The most compelling argument for the adoption of DOTP lies in its environmental and safety profile. DOP belongs to the family of phthalates, some of which have faced increasing regulatory restrictions in the European Union and other regions due to health and environmental concerns. DOTP, being a non-phthalate plasticizer, is not subject to these restrictions. It is widely recognized as an eco-friendly alternative that does not compromise on performance.
Applications
Due to its superior properties, DOTP has found a stronghold in various high-performance applications. It is extensively used in the production of PVC cables rated for 70°C, automotive interiors (helping to reduce fogging on windows), artificial leather, and even in medical applications such as blood storage bags. Its versatility extends to coatings, synthetic rubbers, and as a lubricant additive for precision instruments.
Conclusion
The shift from DOP to DOTP represents more than just a change in chemical composition; it signifies an evolution in material science toward sustainability and higher performance. As industries and consumers alike demand safer and more durable products, DOTP emerges as a prime example of how innovation can meet regulation, delivering materials that are not only effective but also responsible.